|
Rupture of an O-H bond in water requires a very significant (~
490 kJ/mole) input of energy. (The arrow depicting bond rupture
appears with a break inserted in that the energy involved (bond
dissociation enthalpy), is more than an order of magnitude larger
than the 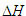 for the overall reaction.)
for the overall reaction.)
|

